
Vendors peddling their wares to bus passengers traveling into northern Uganda
(Photo by Rebecca Sesny)
GULU, Uganda – If it’s not one war, it’s another in this dusty town in northern Uganda. Until a few years ago, Gulu was home to the Lord’s Resistance Army and its leader, Joseph Kony, who terrorized the region – destroying villages, mutilating and killing parents, and forcing children to join his forces.
The Ugandan army finally drove Kony and his rebel forces into central Africa. But a different, more lethal kind of war still devastates Gulu. Families grieving over their losses to Kony are fighting for survival in one of the world’s epicenters of deadly diseases. Foreign donors, medical professionals and pharmaceutical companies have come in to help this remote region, located in the heart of Uganda’s HIV/AIDS zone, but the latest war is creating its own painful divisions.
Past the old rusted tin roofs and mud huts of this region sits a building so modern that it looks out of place. Half a dozen years ago, it housed government troops. It has since become home to the Joint Clinical Research Centre, Gulu Regional Centre of Excellence. The clinic has two roles. It treats thousands of Ugandans with conventional therapies. It also tests new drugs on patients. That makes this clinic part of a booming drug trial business in Uganda, bolstering the government’s struggling health care system.
The dual role creates two tracks at the Gulu clinic – or, as some critics call it, a system of haves and have-nots. Some of the millions of dollars of international support help fund costly treatment for HIV/AIDS patients – not enough, caregivers say. And some of those dollars go to finance drug trials seeking new and better treatments for HIV/AIDS and other diseases.
Trials are closely monitored and fully supplied, according to researchers, creating a dilemma for Ugandan patients: to opt for proven treatments that may be in short supply, or to sign up for a possibly risky clinical trial and at least be assured of a full course of treatments.
Signing up for a trial is voluntary, but that doesn’t make the decision easier – especially for parents who must decide what is best for their children.
“The problem is that inadequate medical care creates a strong impetus for parents to agree to have their kids in research,” said Elizabeth Woeckner, president of Citizens for Responsible Care and Research, an organization that works to protect people who are the subjects of scientific research. “What should be voluntary is not quite so.”
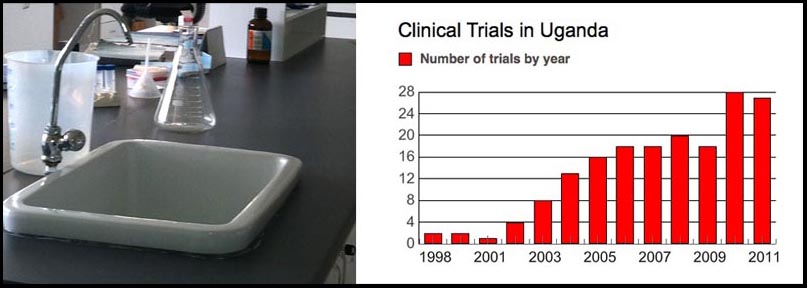
The bars represent the number of pharmaceutical trials started in Uganda during a particular year.
(Photo and Chart by Rebecca Sesny. Source: U.S. National Institutes of Health)
Countless lives are at stake. About 1.4 million Ugandans live with HIV/AIDS, a number that ranks their nation among the world’s top 10 in victims of the disease. Within Uganda, seven out of 100 people in the region around Gulu are infected, one of the highest rates in the country.
All the same, drugs approved for fighting the infection are in short supply at the Joint Clinical Research Centre, say its leaders.
“My work is to make you not miss your drugs,” said Dr. Willy Abongs, who was interviewed in August 2012 when he was the clinic’s director. “But we are operating with drug supplies at 70 percent” of what is needed.
The clinic has had problems getting the proper amounts of the right drugs to patients, Abongs said. Its orders from National Medical Stores, a drug supply company owned by the government and charged with delivering medicine to clinics across the country, consistently fall short.
The lack of resources is possible to see at the Joint Clinical Research Centre. During a reporter’s visit in August, some cabinets were filled with expired medicine waiting to be removed and destroyed. Shelves in the pharmacy were barren.
A shortage of government finances is only one factor keeping the shelves empty. Many medications have ended up in the black market instead of arriving at the clinics that need them most. Other drugs are stolen or lost during overseas shipments and long travel to remote regions, according to caregivers.
Then there are the problems caused by the logistical realities of distributing medicinal drugs in Uganda. “A clinic way up north is not going to get as much as a clinic in the capital, and that’s assuming ideal conditions,” said Woeckner of Citizens for Responsible Care and Research.
Patients have trouble getting to and from clinics, paying for medication, receiving treatments on time, getting tested for a disease, and getting regular blood work done.
Doctors at the Gulu clinic have to split dosages among several patients and wait until the next supply is delivered, Abongs said. Doctors ask patients to come back for the rest of their doses when the drugs are available. Some, however, don’t make the trip back. Getting patients to make medical appointments is a recurring problem, and requiring additional visits because of drug shortages only makes the problem worse.
Lablite, a London-based organization that tracks health issues in African nations, conducted a survey last year that supports what Abongs said. It examined whether HIV/AIDS testing kits and key medications were getting to adults and children in Uganda. The results documented that shortages were “frequent,” with delays for drugs sometimes lasting weeks, and other times several months.
Patients encounter immense difficulty receiving medication. They have trouble getting to and from clinics, paying for medication, receiving treatments on time, getting tested for a disease, and getting regular blood work done.
Those in a drug trial, by contrast, face a much easier regimen. More than a dozen clinical trials are being conducted at the Joint Clinical Research Centre, including Chapas 3, a drug trial for children infected with HIV/AIDS. For all their risks – the drug combinations have not been approved for general use – These trials are touted as the best hope for combating an epidemic ravaging the developing world.
“In a trial kids are followed more carefully and regularly, staff, have generally more time (as funded by trial) and drugs never run out,” said Diana Gibb, trial coordinator of the Medical Research Council, a British group that supports biomedical research. She responded to questions by email.
In trials, medicines remain firmly under the control of the clinicians conducting the tests, Abongs confirmed. “One reason families were happy for their children to be enrolled in a trial is that it was a way to ensure a continuous supply of drugs,” he said.
In addition to adequate drug supplies and regularly monitored patients, there are other perks for those willing to sign up for trials.
“Help for families for transport is provided by the trial,” said Gibb. “We generally have very low drop-out rates. So all possible is done to help families attend with their children, and the trial team keep in contact in other ways as well.”

Among northern Uganda’s afflictions is a mysterious condition known as nodding disease. The thousands of children affected by the problem, whose cause is unknown, suffer seizures that can resemble rapid nodding at the sight of food. The condition leads to mental and physical disabilities. The girl shown in this photo (and in our cover photo), Nancy Lwamwaka, is tied to a tree to prevent her from wandering into danger.
(Photo by Edward Echwalu)
While HIV/AIDS and malaria dominate international registries of drug trials conducted in Uganda, those are not the only diseases subject to pharmaceutical tests. Among others are tuberculosis and meningitis.
In the last five years, drug trials in Uganda have nearly doubled. There were 37 clinical trials in the country before 2007. Since then the number has grown by more than 100 in the last five years.
Records of the U.S. National Institutes of Health show that in 2000, only two new trials were approved for Uganda. But beginning in 2005 Uganda became the fourth most popular site for clinical trials in Africa, behind Egypt, South Africa and Kenya. The same rate of growth also makes Uganda one of the most popular sites for trials among developing countries worldwide.
Drug companies such as Bristol Myers Squibb, Pfizer and Novartis, as well as American agencies, including the National Institutes of Health and the National Cancer Institute, work in places like Uganda because of the low cost and the number of patients who will sign up quickly for tests.
The major drug companies recognize that their research offers benefits and risks to the volunteers under treatment, according to Pharmaceutical Research and Manufacturers of America, an industry group. However, the group said in a statement on its web site, “Medical progress is not possible without human research.”
Along with the promise of solving major health issues, drug trials can bring enormous economic benefit. As a growing source of foreign revenue, clinical trials can mean millions of dollars in spending for developing nations. They also can save money for the companies conducting the tests.
“Conducting clinical trials in low-and-middle-income countries can often reduce overall trial costs by 50 percent and increase the speed of patient enrollment by several fold,” said Thomas Bollyky, chairman of the Center for Global Development, a Washington-based think tank that examines policies affecting developing nations.
Along with economic gains for Uganda come other benefits. Not only are new medicines and vaccines tried out, but local Ugandan health workers can make gains by learning from international researchers. The money brought in also helps make up the gap in much needed health care spending.
“Government budgets for global health are tightening,” said Bollyky, “and new donor funding for delivery is increasingly scarce.”
More than a dozen clinical trials are being conducted at the Joint Clinical Research Centre in Gulu. In Chapas 3, the trial for children infected with HIV/AIDS, parents have to agree to allow HIV/AIDS-infected babies as young as one month old to be part of the study. The testing, which began in 2010, is set to end this year. About 475 children in the central African nations of Uganda and Zambia are enrolled in the long-term experiment, including 55 in Gulu. Those 55 are less than one percent of all the HIV/AIDS-infected patients getting care at the Gulu clinic.
The trial uses two drugs as a control and tests three others: Stavudine, Zidovudine, and Abacivor. Studies have shown that these drugs have adverse side effects in adults, but researchers do not know the impact on children.
Abacivor, a relatively costly drug, can cause severe allergic reactions, including nausea, fever, rash and in some instances death, according to the National Institutes of Health. It is also connected with serious liver problems. Researchers stated they have seen few side effects in African children.
Zidovudine, among other side effects, can cause severe anemia, according to the NIH. That is a major concern for children in Uganda, where malaria, which can also cause anemia, is a serious problem. This HIV/AIDS drug may make the anemia problem worse.
Stavudine is often used in clinical trials across the developing world because of its availability on the inexpensive, generic pharmaceutical market. However, it is known for a condition that can cause severe nerve damage to a patient’s feet, legs and hands.
Despite safeguards, since the late 1990s a number of well-publicized cases have highlighted tests that appeared to violate ethical standards and regulations.
On World AIDS Day in 2009, the World Health Organization called on all countries to phase out the use of Stavudine in HIV/AIDs treatment and research; however, it is still approved by the U.S. Food and Drug Administration, allowing clinical trials receiving U.S. funding to continue the use of Stavudine on patients.
Britain’s Medical Research Council notes that severe side effects from this drug are rare in young children. The council says that its study intends in part to find out when to switch patients from Stavudine to another drug, avoiding such complications.
A drug trial in Uganda or anywhere else that receives any amount of U.S. government funding is subject to oversight. In part, the government uses Institutional Review Boards, or IRBs, which review the ethics of research projects and protect patients’ rights.
A division of the Department of Health and Human Services known as the Office for Human Research Protections determines standards for IRBs. These include requirements that the risk to test subjects be minimized, that all risk be reasonable in relation to anticipated benefit, that the informed consent of prospective test subjects must be legally authorized and that additional safeguards are included to protect vulnerable test subjects.
Despite these safeguards, since the late 1990s a number of well-publicized cases have highlighted tests that appeared to violate ethical standards and regulations.
The Rebecca Project for Human Rights, an organization that advocates for women and girls across the globe, cited several cases of bad drug trials in a report compiled by Dr. David Gisselquist.
“The researchers are effectively unconstrained by U.S. regulations, even through the funding comes from the U.S.,” Gisselquist said. “The public authorities and university researchers have given themselves permission to do unethical research, and who’s to stop them?”
Before a 1997 trial in Uganda funded by the National Institutes of Health for the drug Nevirapine, warnings of toxicity were published by the FDA. These were all but ignored by the research team, according to Gisselquist. During the trial there were serious problems in record keeping and underreporting of life threatening problems, resulting in the deaths of 14 people.
Another study tested the consequences of interrupted treatment. Men were separated into two groups. One continued to receive HIV/AIDS treatment, the other had 12 weeks of treatment and 12 weeks without. After two years the review board found that patients with interrupted treatment had doubled their risk of disease progression, meaning stage four or death. The Rebecca Project alleges that the study pressured participants to remain in the trial, despite the manifest risks.
The Chapas 3 clinical trial in Gulu is set to end this year. At that point, the children enrolled will return to care provided by Uganda’s health system.
The government will do what it can. The Uganda Ministry of Health recently announced a new initiative to decrease mother-to-child transmission of HIV/AIDS infections. The intervention, called Option B+, calls for continuous anti-retroviral treatment for women during and after pregnancy. The Ministry of Health hopes to reduce mother-to-child transmission by 90 percent in the next three years – the kind of major success that Ugandans have long been waiting for. ■
Kamana Shrestha and Alexander Tucciarone contributed to this story.





One Comment